Our experimental and modelling studies aim at understanding the mechanisms involved in the recognition of the olfactory signal and its translation in a behavioural response adapted to the environment and the physiological state of insects.
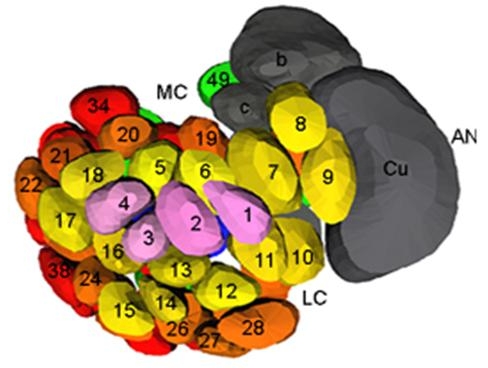
These studies concern the processes of sensory coding, from the chemo-electrical transduction in the olfactory receptor neurones to its integration in the central nervous system by the neuronal network of the antennal lobe as well as the resulting oriented locomotor response.
We also analyse how insects respond to specific signals in a complex sensory environment (interactions of pheromones and plant volatile compounds) and the mechanisms of the modulation of the olfactory response, notably by steroid hormones involved in development.

We use complementary approaches including:
- anatomy (quantitative neuroanatomy, intracellular stainings),
- molecular biology (cloning, in situ hybridization, RNA interference, heterologous expression),
- electrophysiology (extracellular, patch clamp in vivo and in vitro),
- imaging (calcium imaging), ethology (olfactometry, trajectometry, learning assays),
- biochemistry (immuno-dosage of steroids), physico-chemistry (gas chromatography coupled to behaviour)
- and modelling (statistical analyses, computer simulation, robotics) to study the functioning of neurones and neurone networks and modifications of this functioning (global change, pesticides, etc.).
Various olfactometers, locomotion compensators and a wind tunnel are used to record and analyse insect behavior in response to odour signals.
- Our studies are carried out on three species of phytophagous insects.
Our main model is the moth sex pheromone communication with two Noctuidae Spodoptera littoralis and Agrotis ipsilon, because of its sensitivity, specificity and stereotyped behavioural responses involved.
Responses to the pheromone in interaction with plant volatiles are also studied in the context of an applied project, on the invasive weevil Rhynchophorus ferrugineus.

Objectives
- To study and model how the quality, intensity and time pattern of olfactory signals are encoded in the peripheral and central nervous system.
- To decipher and model the signalling pathway of the olfactory transduction.
- To study the mechanisms underlying plasticity linked to the physiological state.
- To analyse interactions between odorants, in particular between pheromone and volatile plant compounds.
- To better estimate the role of the olfactory environment in the modulation of long distance attraction in an applied perspective.

Team news | Team publications | Team members | ↑ top
Team news
Pheromone-mediated command from the female to male clock induces and synchronizes circadian rhythms of the moth Spodoptera littoralis
To extract any adaptive benefit, the circadian clock needs to...
Lire la suiteMeeting of the ADALEP network 2023
On October 24 and 25, the Ecosens department hosted the...
Lire la suiteA Robot to Track and Film Flying Insects
Scientists from the CNRS, Université de Lorraine, and Inrae have...
Lire la suiteTeam news | Team publications | Team members | ↑ top
Team publications
Only applies to publications from 2017 to present. To see all the publications go to the Publications page.
| Référence | Liens | Journal |
|---|---|---|
| SOKOLOWSKI M., BOTTET G., DACHER M.* (2024) Measuring honey bee feeding rhythms with the Beebox, a platform for nectar foraging insects. Physiol Behav, in press. | https://doi.org/10.1016/j.physbeh.2024.114598 🔗 HAL | Physiology & Behavior |
| Maria A.*, Malbert-Colas A~. Braman V.^, Dacher M.*, Chertemps T.*, Maïbèche M.*, Blais C.*, Siaussat D.* 2019 Effects of Bisphenol A on post-embryonic development of the cotton pest, Spodoptera littoralis. Chemosphere 235, 616-625. | 10.1016/j.chemosphere.2019.06.073 🔗 HAL | Chemosphere |
| Hoffmann A.°, Bourgeois T.*, Munoz A.°, Anton S., Gevar J.*, Dacher M.*, Renou M.* (2020) A plant volatile alters the perception of sex pheromone blend ratios in a moth. J Comp Physiol A, 206, 553-570. | 10.1007/s00359-020-01420-y 🔗 HAL | JOURNAL OF COMPARATIVE PHYSIOLOGY A- NEUROETHOLOGY SENSORY NEURAL AND BEHAVIORAL PHYSIOLOGY |
| Fraichard S., Legendre A., Lucas P., Chauvel I., Faure P., Artur Y., Neiers F., Briand L., Ferveur J.-F., Heydel J.-M. (2020) Modulation of sex pheromone discrimination by the UDP-glycosyltransferase Ugt36E1 in Drosophila melanogaster. Genes 11(3). | 10.3390/genes11030237 🔗 HAL | Genes |
| Hostachy C.^°, Couzi P.*, Portemer G.°, Hanafi M.°, Murmu M.^, Deisig N.*, Dacher M.* (2019)Exposure to conspecific and heterospecific sex-pheromones modulates gustatory habituation in the moth Agrotis ipsilon. Frontiers in Physiology, 10: 1518. | 10.3389/fphys.2019.01518 🔗 HAL | Frontiers in Physiology |
| Hostachy C.°^, Couzi P.*, Hanafi-Portier M.°, Portemer G.°, Halleguen A.°, Murmu M.^, Deisig N.*, Dacher M.*, 2019. Responsiveness to sugar solutions in the moth Agrotis ipsilon: parameters affecting proboscis extension. Frontiers in Physiology, 10: 1423. | 10.3389/fphys.2019.01423 🔗 HAL | Frontiers in Physiology |
| Renou, M*, Les insectes ingénieurs. 5. Odeurs et biomimétisme. Insectes,195: 17-21. | Insectes | |
| Conchou L. ^, Lucas P. *, Meslin C. *, Proffit M., Staudt M., Renou M. * (2019) Insect Odorscapes: From Plant Volatiles to Natural Olfactory Scenes.Front. Physiol. 10:972. | 10.3389/fphys.2019.00972 🔗 HAL | Frontiers in Physiology |
| Levakova M., Kostal L., Monsempès C. *, Lucas P. *, Kobayashi R. (2019) Adaptive integrate-and-fire model reproduces the dynamics of olfactory receptor neuron responses in moth. J. R. Soc. Interface 16:20190246. | 10.1098/rsif.2019.0246 🔗 HAL | Journal of The Royal Society Interface |
| Chapuy C.°~, Ribbens L., Renou M.*, Dacher M.*, Armengaud C. (2019) Thymol affects congruency between olfactory and gustatory stimuli in bees. Sci Rep, 9: 7752. | 10.1038/s41598-019-43614-8 🔗 HAL | Scientific Reports |
| Pawson S. M., Kerr J. L., O’Connor B., Lucas P.*, Martinez D., Allison J. D., Strand T. M. (2020) Light weight portable electroantennography device as a future tool for applied chemical ecology. J. Chem. Ecol. 46 :557-566. | 10.1007/s10886-020-01190-6 🔗 HAL | Journal of Chemical Ecology |
| Langlois L., Dacher M.*, Nugent F. (2018). Dopamine receptor activation is required for GABAergic spike timing-dependent plasticity in the ventral tegmental area. Front Syn Neurosci, 10: 32. | 10.3389/fnsyn.2018.00032 🔗 HAL | Frontiers in Synaptic Neuroscience |
| Levakova M., Kostal L., Monsempès C. *, Jacob V. ^, Lucas P. * (2018) Moth olfactory receptor neurons adjust their encoding efficiency to temporal statistics of pheromone fluctuations. PLoS Comput. Biol. 14(11): e1006586. | 10.1371/journal.pcbi.1006586 🔗 HAL | PLoS Computational Biology |
| Aviles A. ~, Boulogne I.^, Durand N.^, Maria A.*, Cordeiro A.°, Bozzolan F.*, Goutte A., Alliot F., Dacher M.*, Renault D., Maïbèche M.*, Siaussat D.* 2019 Effects of DEHP on post-embryonic development, nuclear receptor expression, metabolite and ecdysteroid concentrations of the moth Spodoptera littoralis. Chemosphere 215, 725-738. | 10.1016/j.chemosphere.2018.10.102 🔗 HAL | Chemosphere |
| Chatterjee A*, Lamaze A, De J, Mena W, Chélot E, Martin B, Hardin P, Kadener S, Emery P, Rouyer F. Reconfiguration of a Multi-oscillator Network by Light in the Drosophila Circadian Clock. Curr Biol. 2018 Jul 9;28(13):2007-2017.e4. | 10.1016/j.cub.2018.04.064 🔗 HAL | current biology |
| Machon J.~, Lucas P.*, Ravaux J., Zbinden M. (2018) Comparison of Chemoreceptive Abilities of the Hydrothermal Shrimp Mirocaris fortunata and the Coastal Shrimp Palaemon elegans. Chem Senses 7:489–501. | 10.1093/chemse/bjy041 🔗 HAL | Chemical Senses |
| Jacob V. ^, Monsempès C. *, Rospars J.-P. *, Masson J.-B., Lucas P. * (2017) Olfactory coding in the turbulent realm. PLoS Comput. Biol. 13(12):e1005870. | 10.1371/journal.pcbi.1005870 🔗 HAL | PLoS Computational Biology |
| Bozzolan F*, Durand N^, Demondion E*, Bourgeois T*, Gassias E, Debernard S*.Evidence for a role of oestrogen receptor-related receptor in the regulation of male sexual behaviour in the moth Agrotis ipsilon.Insect Mol Biol26(4):403-413 | 10.1111/imb.12303 🔗 HAL | Insect Molecular Biology |
| Renou M.*, Anton S. (2020) Insect olfaction in a complex and changing world. Current Opinion in Insect Science, 42:xx-yy. | 10.1016/j.cois.2020.04.004 🔗 HAL | Current Opinion in Insect Science |
| Pannequin R., Jouaiti M., Boutayeb M., Lucas P.*, Martinez D. (2020) Lab-on-cables: Automatic tracking of free-flying insects. Science Robotics 5(43):eabb2890. | 10.1126/scirobotics.abb2890 🔗 HAL | Science Robotics |
| Force E.~, Suray C.*, Dacher M.*, Debernard S.*, (2024). Effect of Adult Male Diet on Fertilization and Hatching in an Insect. microPublication Biology. | https://doi.org/10.17912/micropub.biology.001074 🔗 HAL | microPublication Biology |
| De J, Chatterjee A*. (2021) Perception of Daily Time: Insights from the Fruit Flies. Insects. 2021 Dec 21;13(1):3 | 10.3390/insects13010003 🔗 HAL | insects |
| Force E.~, Sokolowski M., Suray C.*, Debernard S.*, Chatterjee A.*, Dacher M.* (2023), Regulation of Feeding Dynamics by the Circadian Clock, Light and Sex in an Adult Nocturnal Insect, Frontiers in Physiology, 14:1304626 | https://doi.org/10.3389/fphys.2023.1304626 | Frontiers in Physiology |
| Force E.~, Couzi P.*, Dacher M.*, Debernard S.* (2023), Diet Impacts the Reproductive System’s Maturation in the Male Moth Agrotis ipsilon (Noctuidae, Lepidoptera), J Insect Physiol, 148:104532. | https://doi.org/10.1016/j.jinsphys.2023.104532 🔗 HAL | J Insect Physiol |
| Li Z.~, Capoduro R.^, Zhang S.~, Sun D., Lucas P.*, Dabir-Moghaddam D.^, François M.C.*, Liu Y., Wang G., Jacquin-Joly E.*, Montagné N.*, Meslin M.*(2023) A tale of two copies: evolutionary trajectories of moth pheromone receptors. Proc Natl Acad Sci USA 120 (20) | https://doi.org/10.1073/pnas.2221166120 🔗 HAL | Proceedings of the National Academy of Sciences of the United States of America |
| Aguilar P., Bourgeois T., Maria A.*, Couzi P.*, Demondion E., Bozzolan F.*, Gassias E., Force E.~, Debernard S.* (2023), Methoprene-tolerant and Krüppel homolog 1 are actors of juvenile hormone-signaling controlling the development of male sexual behavior in the moth Agrotis ipsilon, Horm Behav, 150:105330 | https://doi.org/10.1016/j.yhbeh.2023.105330 🔗 HAL | Horm Behav |
| Zheng Y.~, Cabassa-Hourton C.*, Planchais S.*, Crilat E.*, Clément G., Dacher M.*, Durand N.~, Bordenave-Jacquemin M.*, Guivarc’h A.*, Dourmap C.~, Carol P.*, Lebreton S.*, Savouré A.* (2022). Pyrroline-5-carboxylate dehydrogenase is an essential enzyme for proline dehydrogenase function during dark-induced senescence in Arabidopsis thaliana. Plant Cell and Environment. Online ahead of print. | 10.1111/pce.14529 🔗 HAL | Plant Cell and Environment |
| Vandroux P.°, Li Z.~, Capoduro R.^, François M-C.*, Renou M.*, Montagné N.*, Jacquin-Joly E.* (2022) Activation of pheromone-sensitive olfactory neurons by plant volatiles in the moth Agrotis ipsilon does not occur at the level of the pheromone receptor protein. Front. Ecol. Evol., 10:1035252 | 10.3389/fevo.2022.1035252 | Frontiers in Ecology and Evolution |
| Renou M.* (2022) Is the evolution of insect odorscapes under anthropic pressures a risk for herbivorous insect invasions? Current Opinion in Insect Science. 52: 100926 | 10.1016/j.cois.2022.100926 | Current Opinion in Insect Science |
| Lucas P.*, Anton, S. (2021) Editorial: Invertebrate neurobiology: Sensory Systems, Information Integration, Locomotor- and Behavioral output. Front. Physiol. 12:807521 | 10.3389/fphys.2021.807521 🔗 HAL | Frontiers Physiology |
| Aviles A.~, Cordeiro A.°, Maria A.*, Bozzolan F.*, Boulogne I.^, Dacher M.*, Goutte A.°, Alliot F., Maibeche M.*, Massot M.*, Siaussat D.* (2020) Effects of DEHP on ecdysteroid pathway, sexual behaviour and offspring of the moth Spodoptera littoralis. Hormones Behav, 125: 104808. | 10.1016/j.yhbeh.2020.104808 🔗 HAL | Hormone and Behaviour |
| Frat L~, Chertemps T*, Pesce E, Bozzolan F*, Dacher M*, Planelló R, Herrero O, Llorente L, Moers D, Siaussat D*. Single and mixed exposure to cadmium and mercury in Drosophila melanogaster: Molecular responses and impact on post-embryonic development. Ecotoxicol Environ Saf. 2021 May 27;220:112377. | 10.1016/j.ecoenv.2021.112377 🔗 HAL | ECOTOXICOLOGY AND ENVIRONMENTAL SAFETY |
| Conchou, L^.; Lucas, P*.; Deisig, N*.; Demondion, E.*; Renou, M*. Effects of Multi-Component Backgrounds of Volatile Plant Compounds on Moth Pheromone Perception. Insects 2021, 12, 409. | 10.3390/insects12050409 🔗 HAL | Insects |
| Haouzi, M., Gévar, J*., Khalil, A., & Darrouzet, E. (2021). Nest structures display specific hydrocarbon profiles: insights into the chemical ecology of the invasive yellow-legged hornet Vespa velutina nigrithorax. Chemoecology, 1-12. | 10.1007/s00049-021-00343-7 🔗 HAL | Chemoecology |
| Gassias E, Maria A*, Couzi P*, Demondion E*, Durand N, Bozzolan F*, Aguilar P, Debernard S*. (2021) Involvement of Methoprene-tolerant and Krüppel homolog 1 in juvenile hormone-signaling regulating the maturation of male accessory glands in the moth Agrotis ipsilon. Insect Biochem Mol Biol. 2021 May;132:103566. | 10.1016/j.ibmb.2021.103566 | INSECT BIOCHEMISTRY AND MOLECULAR BIOLOGY |
| RENOU, Michel* ; SORDELLO, Romain ; REYJOL, Yorick, La biologie de la conservation doit-elle prendre en compte les paysages odorants ?, Science Eaux & Territoires | 10.14758/SET-REVUE.2021.HS.03 | Science Eaux & Territoires |
| Renou, M. Mesurer le comportement olfactif des insectes.Insectes, 199:11-15 | Insectes | |
| Murmu M.S.^, Hanoune J.°, Choi A.°, Bureau V.°, Renou M.*, Dacher M.*, Deisig N.*, Modulatory effects of pheromone on olfactory learning and memory in moths. Journal of Insect Physiology, J Insect Physiol 127, 104159. | 10.1016/j.jinsphys.2020.104159 🔗 HAL | Journal of Insect Physiology |
| Gévar, J*., Bagnères, A. G., Christidès, J. P., Darrouzet, E. (2017). Chemical heterogeneity in inbred European population of the invasive hornet Vespa velutina nigrithorax. Journal of Chemical Ecology, 43(8), 763-777. | 10.1007/s10886-017-0874-4 🔗 HAL | Journal of Chemical Ecology |
 All publications of iEES Paris
All publications of iEES Paris Team news | Team publications | Team members | ↑ top
Team members
| Nom Prénom | Corps | Employeur | Adresse | Téléphone | Mél |
|---|---|---|---|---|---|
| CHATTERJEE Abhishek | CR | INRAE | INRAE de Versailles bâtiment 1 – Sous-sol étage – bureau : 17 | (+33) 01-30-83-37-37 | abhishek.chatterjee@inrae.fr |
| DACHER Matthieu | MC | S-U | Campus Pierre et Marie Curie – Paris 5e Tour 44-45 – 3e étage – bureau : 310 | (+33) 01-44-27-65-87 | matthieu.dacher@sorbonne-universite.fr |
| DEBERNARD Stéphane | MC | S-U | Campus Pierre et Marie Curie – Paris 5e Tour 44-45 – 3e étage – bureau : 308 | (+33) 01-44-27-38-39 | stephane.debernard@sorbonne-universite.fr |
| FABRE Caroline | CDD Chercheuse | INRAE, MSCA fellow | INRAE de Versailles bâtiment 1 – RDC – bureau : 6C | caroline.fabre@inrae.fr | |
| FORCE Evan | Doctorant | Univ. Paris-Saclay | Campus Pierre et Marie Curie – Paris 5e Tour 44-45 – 3e étage – bureau : 318 | evan.force@universite-paris-saclay.fr | |
| GHOSH Sagnik | Doctorant | Univ. Paris-Saclay | INRAE de Versailles bâtiment 1 – RDC – bureau : 10 | sagnik.ghosh@inrae.fr | |
| LUCAS Philippe | DR | INRAE | INRAE de Versailles bâtiment 1 – RDC – bureau : 10C | (+33) 01-30-83-37-37 | philippe.lucas@inrae.fr |
Team news | Team publications | Team members | ↑ top